Photos / Sounds
What
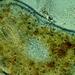
Dicots [Paraphyletic] (Class Magnoliopsida)Observer
zitsermDescription
I thought this was Common mugwort, but that has alternate leaves I think. Not sure what this plant is.
Photos / Sounds
What
Climacia areolarisObserver
wapogipo88Description
There were HUNDREDS of these little cocoons on the wall (rocks) entering the North Woods. Not even 5 feet from the stream. These enchanted and intrigued me so much. Such intricate delicate beauty.
Photos / Sounds
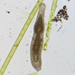
What
Louisiana Long-necked Ground Beetle (Colliuris ludoviciana)Observer
zihaowangDescription
On my balcony. NYC first.
Photos / Sounds
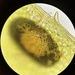
What
Cratomus megacephalaObserver
elaphrornisDescription
Marine Park Salt Marsh, Brooklyn. Burnett Street side. On Iva frutescens.
Photos / Sounds
What
Provancher's Fleabane (Erigeron philadelphicus var. provancheri)Observer
the_buckeye_botanistDescription
Thanks to @mjpapay noticing a funky (note: my word not theirs...) Erigeron at this site in the past (https://www.inaturalist.org/observations/66655862) and making the connection that it could very well possibly be var. provancheri and making me want to investigate further!
After visiting the site and finding 100+ plants growing in boulder crevices and limestone bedrock cracks/ledges, I'm pretty convinced this is indeed var. provancheri. This would make for a considerable range extension of this global rarity and a new state record. Likely to be found along the similar shorelines of nearby Kelleys Island and the Bass Islands, which I plan to survey soon. I believe it should be sought in similar limestone shoreline habitat in Michigan as it stands to reason it would be there, too.
These plants were all quite small, basal leaves/rosettes persistent at/through flowering, the leaves and stems usually pretty glabrate/glabrous (varied plant to plant but some were pretty darn smooth), growing in the specific habitat, and blooming far later than typical philadelphicus would be. They were also staunchly perennials based on the root system and toughness to pull out when collecting a few specimens.
Would love other's opinions who've seen/experienced this plant before.
Photos / Sounds
What
Bottlebrush Frost Lichen (Physconia detersa)Observer
stephen_bucklinDescription
No lichen substances. White medulla. Squarrose rhizines.
Photos / Sounds
What
Yellow Pouncewort (Lejeunea flava)Observer
polarblairxDescription
Underleaves large, most over half the size of lateral leaves. Free margin of lobules inrolled.
Found growing on bark in an orchid pot in the NYBG gift shop
Photos / Sounds
What
Vankya heufleriObserver
sus_scrofaDescription
full credit for finding and putting in the work to identify this plant pathogen goes to Andrew Cannon.
on underside of leaf of Erythronium americanum
Photos / Sounds
What
Grove Earwort (Scapania nemorea)Observer
zihaowangDescription
Gemmae unicellular; dorsal lobe of leaf extends over and across the stem
Photos / Sounds
Observer
ethancrensonDescription
On a small hardwood branch with the bark beginning to fall away. Note the white lesions in the bark and wood beneath. Perithecia up to 1mm wide by 1.5mm tall.
Asci: 153-168 x 7.5-10µm. The spore bearing part 105-125µm. IKI+, the apical plug about 2.5 tall x 3µm wide when mature; much longer in immature asci.
Spores: 13-14 x 6-7µm, brown, ellipsoid to inequalateral ellipsoid, guttulate, usually with one large guttule at the center, with a very inconspicuous germ slit less than spore length.
Photos / Sounds
What
Polychaete Worms (Class Polychaeta)Observer
zihaowangDescription
200x and 800x in brackish water.
Photos / Sounds
What
Gastropods (Class Gastropoda)Observer
zihaowangDescription
800x and 200x in sea water. It has a spiral shell. The cilia on the two wheels were actively moving.
Photos / Sounds
Observer
dms1535Description
Aspidiophorus paradoxus is the largest Aspidiophorus species with about 300µm length.
Fig. 1: Dorsal scales.
The entire animal is covered with relatively large rhombic peduncle scales.
Fig. 2: Cross section
In cross-section, the structure of the peduncle scales can be clearly seen: the scales sit with a small basal plate on the cuticle of the animal, from which a thin, hollow peduncle rises. At the end of the peduncle sits a rhombic terminal plate, with a central keel. At the posterior end of the animal, the terminal plates of the last row of scales are enlarged.
The pharynx of the animal is terminally swollen, and the head is weakly five-lobed with two separate pairs of palpal tufts.
Fig. 3: Ventral view.
Ventrally, the strong hypostomion behind the mouth opening is striking. The two ciliated bands split at the head, but the inner branches do not unite in the population I examined. The base of the toes does not bear scales, the adhesive tubes
measure about 50% to 70% of the toe length and taper to a point.
Let's take a closer look at the scales:
Fig. 4: Back scales
In the scales on the back, the rhombic shape of the end plates can be seen most clearly. Less conspicuous - but typical for the species - is the middle keel of the scales.
Fig. 5: Cross section of scales
In cross section the complex geometry of the peduncle scales becomes clear - base plate, peduncle and end plate form a very flexible and stable carapace. The additional cavity under the outer scales acts like a "crumple zone" and further increases the protective effect.
Fig. 6: Cross-section of scale stems.
The stems of the scales consist of hollow tubes that ensure maximum stability with minimum material input - a fascinating example of evolutionary optimization.
The abdomen of the animals is not completely covered with petiolar scales. Rather, they end in the anal region and are joined by simple small, rounded keel scales that are not an obstruction to feces.
Fig. 7: Scaling of the abdomen.
At the furca base some (according to literature 4) spines protrude into the toe cutout.
The head is almost completely covered with slightly smaller peduncle scales:
Fig. 8: Head scales
Cephalion and pleurae are quite small and inconspicuous.
According to literature A. paradoxus has three teeth in the pharynx:
Fig. 9: Mouth armament.
In the animals I examined, only a curved stylet brace was found, the tip of which protrudes into the lumen of the pharynx and probably serves to open algal cells that are conveyed past it. It is possible that the literature reference to "three teeth" is merely based on a microscopic artifact, as the entire clasp may not be in the focal plane as a whole.
Photos / Sounds
What
Genus ChlamydodonObserver
peptolabDescription
Chlamydodon species consistent with C. exocellatus
Sample from the road end at Harbor View Lane where there is a marine channel between Gardiner's Bay and Three Mile Harbor. The cyrtophorid ciliate genus Chlamydodon is characterized by a unique cross-striated band (CSB) along the cell perimeter. To date, more than 15 nominal species have been assigned to this genus, all of which are exclusively from marine or brackish water" (1).
My ciliate was very fat and colorful in that the cytoplasm was literally stuffed with ingested food: green algae and larger red globules in a background of smaller golden yellow globules. Being so well fed, it was impossible to see much internal detail. The cyrtos with toothed nematodesmal rods surrounding the cytostome was not visible nor was the anterior pigment spot and the peripheral cross striated band characteristic of the genus was only partially visible. I took a couple of videos and placed the slide in a humid chamber to allow the little fellow to digest and egest some waste.
The next morning much more morphological detail was visible. The ciliate was now much slimmer and most of the ingested food was gone though the cytoplasm was still full of golden yellow granular material. I could see that this 212 um long ciliate has a continuous cross striated band. The cyrtos consists of 19 toothed nematodesmata. There are in excess of 40 vacuoles scattered all over the body, 5 that are somewhat larger than the more numerous small ones. There is a central ovoid macronucleus. Strangely, I still could not appreciate the characteristic yellow or purple anterior pigment spot.
The above parameters are somewhat consistent with Chlamydodon bourlandi Qu et al 2018 but perhaps more closely resemble another nominal species, C. exocellatus Ozaki & Yagiu, 1941, Snoeyenbos-West et al., 2004. "Chlamydodon bourlandi sp. nov. has a cell size of 150–250 × 65–150 µm, a complete CSB, a reddish to violet cell colour, 83–97 somatic kineties, and 40–68 contractile vacuoles" (1). Qu et al consider Chlamydodon exocellatus sensu Snoeyenbos-West et al., 2004, to be an earlier population of C. bourlandi (1). The former has only 5 contractile vacuoles whereas C. bourlandi has >40. My Chlamydodon has 5 larger vacuoles clustered toward the anterior end of the cytosome in addition to the numerous much smaller vacuoles. Perhaps these smaller ones are the empty food vacuoles created during the overnight diet I inflicted on the ciliate and should not be counted as contractile vacuoles.
Qu et Al state: “Chlamydodon exocellatus sensu Snoeyenbos-West et al., 2004 resembles the new species (C. bourlandi) based on the cell size in vivo (150–230 mm vs. 165–215 mm in the present isolate), the number of somatic kineties (83–97 vs. 87–97), and the number of nematodesmal rods (14–19 vs. 14–17). Although there was no mention of the number of contractile vacuole, we think C. exocellatus sensu Snoeyenbos- West et al., 2004 was misidentified and it should be an early population of the C. bourlandi sp. nov. However, these two populations share a low similarity (94.1%) of the SSU rRNA gene sequences” (1).
Gong et al (2) point out that C. exocellatus lacks an anterior pigment spot like my Chlamydodon and none is depicted by Snoeyenbos-West et al., 2004 (1-fig 45) while Qu et al (1-figs.43 and 44) ) show that C. bourlandi has a purplish pigment spot. Qu et al also show that C. bourlandi also has a ridge from cytostome to posterior end; two grooves longitudinally positioned on dorsal side, on both sides of ridge which I do not see in my Chlamydodon. C. exocellatus can be distinguished from C. bourlandi by a rounded body shape with both ends broadly rounded vs. anterior end widely rounded and posterior end obviously narrowed to pointed in C. bourlandi (1). Finally, C. bourlandi has a purple coloration while my Chlamydodon has a golden yellow coloration.
Thus, I conclude that my observation is most likely C. exocellatus. Imaged in Nomarski DIC using Olympus BH2S under SPlan 40x objective plus variable phone cropping on Samsung Galaxy S9+. Some screen caps further cropped.
1. Zhishuai Qu, Lifang Li, Xiaofeng Lin, Thorsten Stoeck, Hongbo Pan, Khaled A. S. Al-Rasheid & Weibo Song (2018) Diversity of the cyrtophorid genus Chlamydodon (Protista, Ciliophora): its systematics and geographic distribution, with taxonomic descriptions of three species, Systematics and Biodiversity, 16:5, 497-511, DOI: 10.1080/14772000.2018.1456493
2. Jun Gong, Weibo Song, and Alan Warren. Updating the Ciliate Genus Chlamydodon Ehrenberg, 1835, with Redescriptions of Three Species (Ciliophora: Cyrtophorida). Acta Protozool. (2005) 44: 19 - 32. https://www.researchgate.net/publication/236839424
Photos / Sounds
What
Houghton's Sedge (Carex houghtoniana)Observer
taylorsturmDescription
rhizomatous; vegetation glabrous; perigynia sparsely pubescent; plants with red basal sheaths; perigynia ascending, 5mm+ with distinct straight two-tipped beak; perigynia strongly veined (not obscured by pubescence); growing in disturbed sandy opening in pine forest alongside Comptonia peregrina, etc. some individuals had interesting red coloration on perigynia
Photos / Sounds
What
Intestinal Bilharzia Fluke (Schistosoma mansoni)Observer
lauramahechasaracuragcbDescription
Macho adulto
Photos / Sounds
What
Lacrymaria marinumObserver
peptolabDescription
Lacrymaria marina Kahl, 1933 syn. Lacrymaria olor var. marina (O.F. Müller, 1786) Kahl, 1933
It's sexy time for the marine Lacrymaria. Last night I was looking at a slide of my latest sample from the intertidal benthos of marine estuary Accabonac Harbor which had an interesting polychaete and numerous Lacrymaria. I put the slide in a moist chamber overnight and when I reexamined it this morning I found a veritable sex orgy of conjugating pairs dancing their sensual ballet while exchanging genetic material through their mouths. There were equal sized pairs and unequal pairs where one was much smaller. One such unequal pair had the larger partner undergoing transverse fission while conjugating with a small partner 1/4 the size of the larger partner. Another uneven pair had globular bodies and the larger partner had a massively enlarged broad neck. Some pairs got tired and both went into the contracted resting state characteristic of Lacrymaria. Fascinating stuff. Imaged in Nomarski DIC using Olympus BH2 under SPlan 40x and 20x objectives plus phone cropping on Samsung Galaxy S9+.
A lot of images here to show all aspects of the sex orgy. We start with a teaser GIF of conjugation of two normal individuals. Then an illustration from the supporting reference and a picture of the sample site. Then some screen captures of a typical individual. Then screen caps of a fissioning individual. Then screen caps of conjugating pairs in various metabolic states- two equal sized individuals, two semi-resting individuals, two bizarely shaped individuals, and finally one normal individual conjugating with one undergoing transverse fission! Finally, I show GIFs of the various types of conjugating pairs in the same order as the screen caps. A lot of stuff!
Lacrymaria marina Kahl, 1933
Diagnosis. Body size 150-300 × 20-25 μm in vivo; extended spindle to vase-like shape; highly contractible neck; cell colorless; cortical granules colorless, regularly
arranged in between somatic kineties; single contractile vacuole, conical shape and located terminally; papillary head with obliquely arranged oral ciliary rows, 10-12 in number; spiral somatic kineties on contracted body, 17- 20 somatic kineties; 1 ellipsoidal macronucleus. Distribution. China, Germany, Korea (this study).
Remarks. Lacrymaria marina Kahl, 1933 can be separated from L. olor (Müller, 1786) Bory de St. Vincent, 1824 by the number of macronuclear nodules (1 vs. 2),
and habitat (saline water vs. freshwater) (Kahl, 1930). Lacrymaria marina can be separated from L. nana (Vuxanovici, 1961) Song and Wilbert, 1989 by the number of somatic kineties (15-20 vs. about 13), the shape of contractile vacuole (conical vs. spherical), and the position of the contractile vacuole (terminal vs. subterminal) (Song and Wilbert, 1989).
Above descriptions from:
Brief descriptions of 12 ciliate species previously unrecorded (Protozoa: Ciliophora) in Korea. Ji Hye Kim and Jae-Ho Jung. Journal of Species Research 6(Special Edition):15-25, 2017. https://www.researchgate.net/publication/322212723
From Bruce Taylor: From saltwater, with a single macronucleus, posterior vacuole, approximately triangular (possibly a defecatory organelle, per Song & Packoff). Apparently somewhat less extensile than L. filiformis (a freshwater species), bigger than L. nana, lacking the tail-like process of L. acuta.
L. marinum Kahl, 1933 is the same taxon as L. olor var. marina (O.F. Mueller, 1786) Kahl, 1933.
What
Quillworts (Genus Isoetes)Observer
awkwardsDescription
I have been hoping to see this for a while, but am still not sure if it actually is Isoetes. There are about ten.
Photos / Sounds
What
Eastern Fairy Shrimp (Eubranchipus holmanii)Observer
zitsermDescription
Female. In a vernal pool in deciduous woods
Photos / Sounds
What
Genus OctosporaObserver
polarblairxDescription
Found amongst Ceratodon purpureus, Streblotrichum convolutum, Funaria hygrometrica, Gemmabryum, and Bryum argenteum
Photos / Sounds
What
Asa Gray's Scalewort (Frullania asagrayana)Observer
northeast_naturalistDescription
Wider overlapping lobes. Row of differentiated ocelli, “bucket” shaped lower leaves evident under scope. Growing on outcrop of Shawangunk conglomerate.
Photos / Sounds
What
Featherfoil (Hottonia inflata)Observer
soilsflutterbyDescription
I could be wrong, but gosh, i hope I am right.
Photos / Sounds
What
Genus UleiotaObserver
sus_scrofaDescription
found under bark of a hardwood log, possibly an oak.
What
Southern Waternymph (Najas guadalupensis)Observer
pawelpDescription
Took a cutting to grow out in an aquarium and identify
Photos / Sounds
Observer
ethancrensonDescription
These aren't cup-shaped apothecia, but rather collabent (collapsed) pseudothecia in a brown subiculum.
Asci clavate, 76-90 x 13-16µm, containing 32 spores (by my approximation).
Spores hyaline, allantoid:
7.7-12.5 x 1.6-2.4µm
Me 9.3 x 2.2µm
Q=3.5-6.2
MeQ=4.3
N=23
Photos / Sounds
What
Eastern Haircap Moss (Polytrichum pallidisetum)Observer
syrinxedDescription
High Point State Park, on rocks near the monument. I def think it's a Polytrichum.... The capsules seem too long to be commune/juniperum, and they also seem to lack the required disk. Doesn't feel like it's commune in general. The capsules for pallidisetum are described as tan, but seem red in LOTS of photos, esp the one in the Princeton Field Guide to Mosses, and many of the traits match descriptions within said field guide (such as red tipped leaves; boxy & bent over capsules). Actual experts pls help
Photos / Sounds
What
Pensacola Hawthorn (Crataegus lacrimata)Observer
aidancamposDescription
In opening of scrubby Pinus palustris dominated flatwoods.
Photos / Sounds
What
Plumatella Moss Animal (Genus Plumatella)Observer
polarblairxDescription
In organic detritus from many locations, found on plagiomnium undulatum leaf, origin unknown. Statoblast
What
Half-smooth Odostome (Boonea seminuda)Observer
stephanie_nycDescription
These were found in the drift. Because they're so tiny I hadn't realized two of them still had the operculums attached - if they were still alive I would have put them back in the water.
Photos / Sounds
What
Lesser Notchwort (Isopaches bicrenatus)Observer
gpalermoDescription
Aka Lophozia bicrenata.
Growing on sandy soil by the side of a woodland trail.
@zihaowang
The Hepaticae and Anthocerotae of North America by Rudolf Schuster (vol. 2, 1969) describes this liverwort as “An extraordinarily widespread species…with truly ‘weedy’ propensities,” but this observation of Isopaches bicrenata is only the fifth recorded in iNaturalist for all of North America. Although common, it’s inconspicuous and apparently unnoticed.
Photos / Sounds
What
Common Dandelion (Taraxacum officinale)Observer
polarblairxDescription
What a weirdo!
Photos / Sounds
What
Small Spikerush (Eleocharis parvula)Observer
gpalermoDescription
Brackish pond shore exposed by evaporation. Floral scales <2mm long. Stems thread-like, less than 1 mm in diameter. J-shaped to U-shaped tubers. M. Arsenault et al., Sedges of Maine 2013. Third iNaturalist observation of this species in New England.
Photos / Sounds
What
Onion Vine (Paederia cruddasiana)Observer
douggoldmanDescription
Weedy, invasive vine with smelly leaves.
Photos / Sounds
What
Memorial Rose (Rosa lucieae)Observer
shuysmanDescription
On dune on side of paved pathway. Stems prickle-less. Rachis with small recurved thorns
Photos / Sounds
What
Bushy Aster (Symphyotrichum dumosum)Observer
elaphrornisDescription
Marine Park, Brooklyn. Sidewalk space, with Mugwort etc.
Photos / Sounds
What
Bog Smartweed (Persicaria setacea)Observer
wernerehlDescription
A known population. First iNat posting on the 13 state northeastern region. This is the northeastern-most population in the U.S. I noted no peppery taste in the leaves. This is a sturdy perennial with rhizomes, up to about three feet tall but can bloom at 12 inches as well. State-threatened species.
Photos / Sounds
What
Bug-on-a-Stick (Buxbaumia aphylla)Observer
polarblairxDescription
Growing on clay soil on the vertical face of a 6 inch overhang on top of a mudshale cliff next to the raritan. What a find!
Photos / Sounds
What
Orange Polypore Mould (Hypomyces aurantius)Observer
ethancrensonDescription
On Trametes versicolor. Turns dark purple in KOH, then fades to red. Ascospores warted, 1-septate, apiculate with pointy projections, 16-25 x 4-5.5µm. Conidiospores broadly ellipsoid, 1-septate, 12-15 x 7.5-8µm.
Photos / Sounds
What
Frosted Grain-spored Lichen (Sarcogyne regularis)Observer
zacaDescription
Maybe this is the first time I face it; However, not many doubts about the classification; the practically inexistent thallus, the pruinose bluish apothecia growing on siliceous rock (granite) are distinctive. I may have a sample and I will try to confirm the ID soon.